xxxxxThe English chemist and physicist Michael Faraday began working for the eminent scientist Humphry Davy in 1812. Elaborating on the work of the Danish physicist Oersted and the French physicist Ampère, he made the first primitive electric motor in 1821, producing a source of power by transforming electrical energy into mechanical energy. Then in 1831 he demonstrated the principle of electromagnetic induction (the reverse of Oersted’s finding) and explained the laws that governed it. From this discovery he made the first electric dynamo, the forerunner of the modern generator. In chemistry he is especially remembered for his contribution to electrolysis, a method of splitting up a chemical compound by means of an electric current. This had been pioneered by Davy, but Faraday developed the method much further, and in 1834 arrived at its two fundamental laws. He was elected to the Royal Society in 1824, and became professor of chemistry at the Royal Institution when Davy retired in 1833. A man of extraordinary ability, he wrote major works on physics and chemistry, and gained much popularity and renown for his series of lectures, some of them given especially for children.
MICHAEL FARADAY 1791 -
(G3b, G3c, G4, W4, Va, Vb)
Acknowledgements
Faraday: by the English portrait photographer John Watkins (1823-
 xxxxxThe English chemist and physicist Michael Faraday, by his pioneering experiments in electromagnetism, made the first primitive but practical models of the electric motor and dynamo. By so doing he laid the foundations of modern electrical engineering. He also introduced lines of magnetic force, investigated the effect of magnetism on light, and, by his contribution to chemistry -
xxxxxThe English chemist and physicist Michael Faraday, by his pioneering experiments in electromagnetism, made the first primitive but practical models of the electric motor and dynamo. By so doing he laid the foundations of modern electrical engineering. He also introduced lines of magnetic force, investigated the effect of magnetism on light, and, by his contribution to chemistry -
xxxxxFaraday was born in Newington, Surrey, the son of a blacksmith. He had very little formal education, and at the age of fourteen began work as an apprentice to a London bookseller and binder. Over the next seven years he read widely, and became particularly interested in the current books and articles on science. He began experimenting with electricity in his spare time, and at the age of twenty he attended a number of lectures given by Humphry Davy. Afterwards, he sent the notes he had made to this eminent English scientist, and these clearly made an impression. In 1812 he was appointed Davy’s secretary and, later, his laboratory assistant at the Royal Institution. Such was his contribution to the scientific work being carried out there, that he was elected to the Royal Society in 1824, and became the director of the Institution’s laboratory the following year. Then in 1833 when Davy retired, he succeeded him as professor of chemistry. For much of his career, from 1825 to 1862 in fact, he gained a reputation for the popular lectures he gave at the Institution. These included his “Friday evening discourses”, attended on one occasion by Prince Albert and two of his sons. He died at his home near Hampton Court in Surrey, and was buried in Highgate Cemetery in north London.
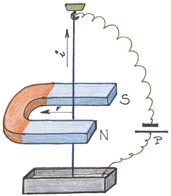 xxxxxIt is in the field of physics that Faraday’s work is best remembered and valued. As we have seen, it was in April 1820 (G4) that the Danish physicist and chemist Hans Christian Oersted discovered that electric current in a wire was able to deflect a magnetic needle. Soon afterwards the French physicist and mathematician André Ampère elaborated on this finding, and demonstrated that magnetic force was a circular force, producing in effect a cylinder of magnetism around the wire. Faraday was the first to realise that this force could be put to work. He reasoned that if a magnetic pole could be isolated, then it would move constantly in a circle around a current-
xxxxxIt is in the field of physics that Faraday’s work is best remembered and valued. As we have seen, it was in April 1820 (G4) that the Danish physicist and chemist Hans Christian Oersted discovered that electric current in a wire was able to deflect a magnetic needle. Soon afterwards the French physicist and mathematician André Ampère elaborated on this finding, and demonstrated that magnetic force was a circular force, producing in effect a cylinder of magnetism around the wire. Faraday was the first to realise that this force could be put to work. He reasoned that if a magnetic pole could be isolated, then it would move constantly in a circle around a current-
xxxxxOver the next ten years his research led him to the conclusion that, since an electric current could cause a magnetic field, a magnetic field should be able to produce an electric current. In 1831 he carried out a series of experiments which demonstrated this principle of electromagnetic induction, (the converse of Oersted’s finding), and explained the laws which governed it. The device he then built to reach this conclusion proved to be the first successful attempt at the construction of an electric dynamo, the forerunner of the modern generator. The following year he illustrated the lines of electric and magnetic force by the use of iron filings. This was seen as unnecessary by most scientists at that time, but later, as we shall see, the Scottish physicist James Clerk Maxwell accepted this concept and put Faraday’s ideas into mathematical form.
 xxxxxFaraday’s advances in chemistry were just as impressive. In this field his greatest contribution was in electrolysis. This method of decomposing or splitting up a chemical compound by passing an electrical current through it had been pioneered by Davy, but Faraday made much greater use of this phenomenon, and in 1834 arrived at its two fundamental laws. Earlier, in the 1820s, he discovered two new chlorides of carbon, and was the first to liquefy chlorine, later using this coolant to liquefy a number of common gases. And during this period -
xxxxxFaraday’s advances in chemistry were just as impressive. In this field his greatest contribution was in electrolysis. This method of decomposing or splitting up a chemical compound by passing an electrical current through it had been pioneered by Davy, but Faraday made much greater use of this phenomenon, and in 1834 arrived at its two fundamental laws. Earlier, in the 1820s, he discovered two new chlorides of carbon, and was the first to liquefy chlorine, later using this coolant to liquefy a number of common gases. And during this period -
xxxxxFaraday, a man of extraordinary ability over a wide range of subjects, astonished the scientific world by his findings, particularly those achieved in physics. Like Davy, he received a number of high honours, including the Royal and Rumford medals of the Royal Society. However, an extremely modest man, he declined the presidency of the society and rejected a knighthood, wishing to remain plain Mr. Faraday. Towards the end of his life he was awarded a pension, and accepted the use of a cottage near Hampton Court. The importance of his contribution to scientific knowledge was immense. It was largely upon the foundation laid down by his painstaking experimental research that the Scottish physicist James Clerk Maxwell was able to build his concept of the electromagnetic field, paving the way for Albert Einstein’s theory of relativity. Apart from papers and articles, Faraday’s major works were Chemical Manipulation of 1827, Experimental Researches in Electricity, three volumes produced from 1844 to 1855, and Experimental Researches in Chemistry and Physics, published in 1859.
 xxxxxIncidentally, between the years 1827 and 1860 Faraday gave nineteen “Christmas Lectures” at the Royal Institution, each “adapted to a juvenile audience”. These lectures for young people are still held every year, and they are named after him. One of the last lectures he gave in this series was entitled The Chemical History of a Candle, and this was the name he gave to the book he published in 1860. Containing six of his children’s lectures, it has become a classic of science literature. ……
xxxxxIncidentally, between the years 1827 and 1860 Faraday gave nineteen “Christmas Lectures” at the Royal Institution, each “adapted to a juvenile audience”. These lectures for young people are still held every year, and they are named after him. One of the last lectures he gave in this series was entitled The Chemical History of a Candle, and this was the name he gave to the book he published in 1860. Containing six of his children’s lectures, it has become a classic of science literature. ……
xxxxx…… A deeply religious man, when the Crimean War broke out in 1853 he was asked to take charge of a project to produce poison gas for use on the battlefield, but he refused to do so. Likewise, when Queen Victoria invited him to lunch on a Sunday, he eventually accepted, but later sought forgiveness from the Church authorities for not attending a service on that day. ……
xxxxx…… Not until he gained universal fame did Faraday overcome his humble origins. When he went on a grand tour of Europe with Davy in 1813, acting as his secretary, Davy’s wife made him sit and eat with the servants.

xxxxxAmong Faraday’s close friends was the prominent Irish physicist John Tyndall (1820-
xxxxxIncidentally, in 1872-

 xxxxxThe English physicist and philosopher William Whewell (1794-
xxxxxThe English physicist and philosopher William Whewell (1794-
xxxxxAnd scientists were also at work abroad. As we have seen, the French mathematician and physicist Siméon-
 xxxxxMeanwhile in the United States it became evident that the brilliant physicist Joseph Henry (1797-
xxxxxMeanwhile in the United States it became evident that the brilliant physicist Joseph Henry (1797-
xxxxxIncidentally, when Henry became the first secretary of the Smithsonian Institution in Washington D.C. in 1846, he began organizing a voluntary network of stations equipped with meteorological instruments. These telegraphed weather reports to the Institution, and these weather conditions were then published. By 1860 about 500 stations were sending in reports, and this led to the founding of the US Weather Bureau in 1870.
 xxxxxIt was around this time (1833) that the Scottish chemist Thomas Graham (1805-
xxxxxIt was around this time (1833) that the Scottish chemist Thomas Graham (1805-
xxxxxGraham was born in Glasgow, the son of a textile manufacturer. He studied in his home town and then at Edinburgh, and spent the years 1837-
 xxxxxAnd it was in this period, in 1827 in fact, that the German chemist Friedrich Wohler (1800-
xxxxxAnd it was in this period, in 1827 in fact, that the German chemist Friedrich Wohler (1800-
xxxxxWohler was born near Frankfurt-

xxxxxIt was while making a study of cyanates that Wohler struck up a close friendship and productive partnership with his compatriot, the German chemist Justus von Liebig (1803-

xxxxxLiebig was born in Darmstadt, Hesse, and studied at Bonn, Erlangen and Paris. He was appointed professor of chemistry at the University of Giessen at the age of 23 and over the next twenty five years made his research laboratory internationally famous, both for the quality of its work in physiological chemistry, and for the string of able scientists it produced. He was appointed to the chair of chemistry at Munich University in 1852. He founded the journal Annals of Pharmacy in 1832, and published his Animal Chemistry ten years later. He was made a baron in 1845.
Including:
John Tyndall, William Whewell,
Joseph Henry, Thomas Graham,
Friedrich Wohler and Justus von Liebig

W4-