


 xxxxxThe German mathematician and physicist Rudolf Clausius
is often credited with the creation of the science of
thermodynamics. He was certainly one of the principal founders of
this branch of science. He was born at Koslin, Prussia (now in
Poland) and, after attending Berlin and Halle universities, was
appointed professor of physics at the Artillery and Engineering
School at Berlin in 1850. It was in that
year that he produced the first outlined statement of the second
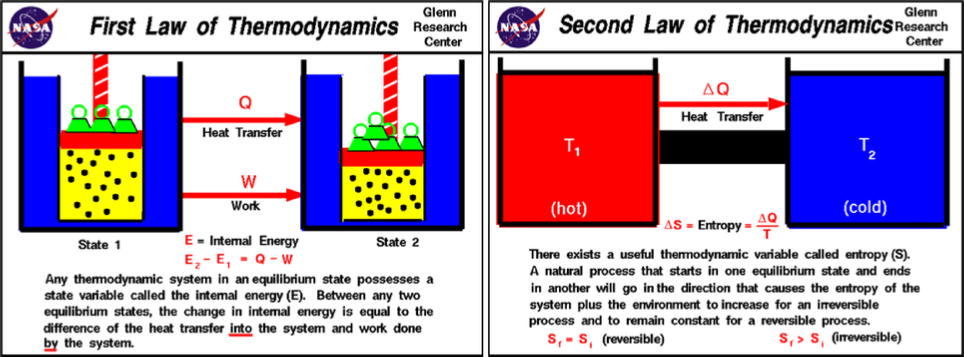
law of thermodynamics, a law which, simply put, states that heat
cannot of itself pass from a colder to a hotter body (see diagram below). He then
went on to apply the laws of thermodynamics to the theory of the
steam engine, and formulated thereby the concept of entropy, i.e.
the measurement of a system’s energy which is not
available for conversion into mechanical work.
xxxxxThe German mathematician and physicist Rudolf Clausius
is often credited with the creation of the science of
thermodynamics. He was certainly one of the principal founders of
this branch of science. He was born at Koslin, Prussia (now in
Poland) and, after attending Berlin and Halle universities, was
appointed professor of physics at the Artillery and Engineering
School at Berlin in 1850. It was in that
year that he produced the first outlined statement of the second
law of thermodynamics, a law which, simply put, states that heat
cannot of itself pass from a colder to a hotter body (see diagram below). He then
went on to apply the laws of thermodynamics to the theory of the
steam engine, and formulated thereby the concept of entropy, i.e.
the measurement of a system’s energy which is not
available for conversion into mechanical work.
xxxxxHe was appointed professor of physics at the Polytechnic Institute in Zurich in 1855, and on his return to Germany in 1867 became professor of physics at Wurtzburg and then at Bonn two years later. From 1857 onwards he conducted major research into the kinetic theory of gases and made important advances in the theory of electrolysis.
RUDOLF JULIUS EMANUEL CLAUSIUS
1822 -
Acknowledgements
Clausius: date
and artist unknown. Diagrams: by
courtesy of NASA Glenn Education, Glenn Research Center,
Cleveland, Ohio, USA, see Beginner’s Guide to Aeronautics,
http://www.grc.nasa.gov/WWW/k-
Including:
James Prescott Joule

Va-
 xxxxxResearchxinto thermodynamics
had been carried out just a few years earlier by two other German
physicists. Jules Robert Mayer (1814-
xxxxxResearchxinto thermodynamics
had been carried out just a few years earlier by two other German
physicists. Jules Robert Mayer (1814-

xxxxxMeanwhile the
English physicist James Prescott Joule (1818-
xxxxxOver a
lifetime devoted to scientific research Joule made a valuable
contribution in the field of thermodynamics. In 1840, in his paper
On the Production of Heat by Voltaic
Electricity, he concluded 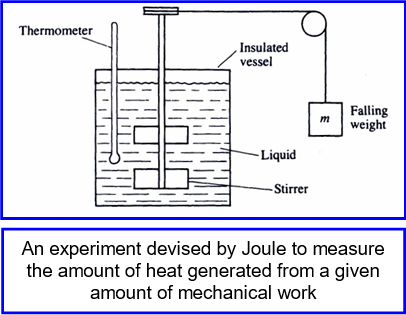 (Joule’s Law) that the heat produced in a
wire by an electric current is proportional to the product of the
resistance of the wire and the square of the current. Then three
years later, after a series of ingenious experiments, he
determined the amount of energy needed to produce a unit of heat (Joule’s equivalent). Later, in the early
1850s, he worked with the Irish physicist William Thomson (later
Lord Kelvin) and produced what came to be known as the “Joule-
(Joule’s Law) that the heat produced in a
wire by an electric current is proportional to the product of the
resistance of the wire and the square of the current. Then three
years later, after a series of ingenious experiments, he
determined the amount of energy needed to produce a unit of heat (Joule’s equivalent). Later, in the early
1850s, he worked with the Irish physicist William Thomson (later
Lord Kelvin) and produced what came to be known as the “Joule-
xxxxxIncidentally, in recognition of his contribution to thermodynamics, a unit of energy was named after him (the Joule), and the letter J was used to represent the value of the mechanical equivalent of heat.


